About MDIAS
What is MDIAS?
The Medical Device Information Analysis and Sharing (MDIAS) initiative is a voluntary partnership between the Food and Drug Administration (FDA) and the private sector. As part of the Case for Quality Collaborative Community (CfQcc) MDIAS was formed to proactively analyze broad and extensive medical device-related data to improve healthcare outcomes for patients.
The concept stems from the recognition that data from a wide variety of sources can provide greater insights than data from a single source alone. MDIAS enables the medical device community to acquire, integrate, and analyze multiple data sources in a way that provides new insights into systemic quality issues.
MDIAS Vision
MDIAS will be trusted as a leading collaborator in continuous improvement for medical device quality, safety, and patient experience.
MDIAS Mission
MDIAS improves patient safety, quality, and satisfaction outcomes by collaboratively leveraging data across the medical device ecosystem.
MDIAS Process
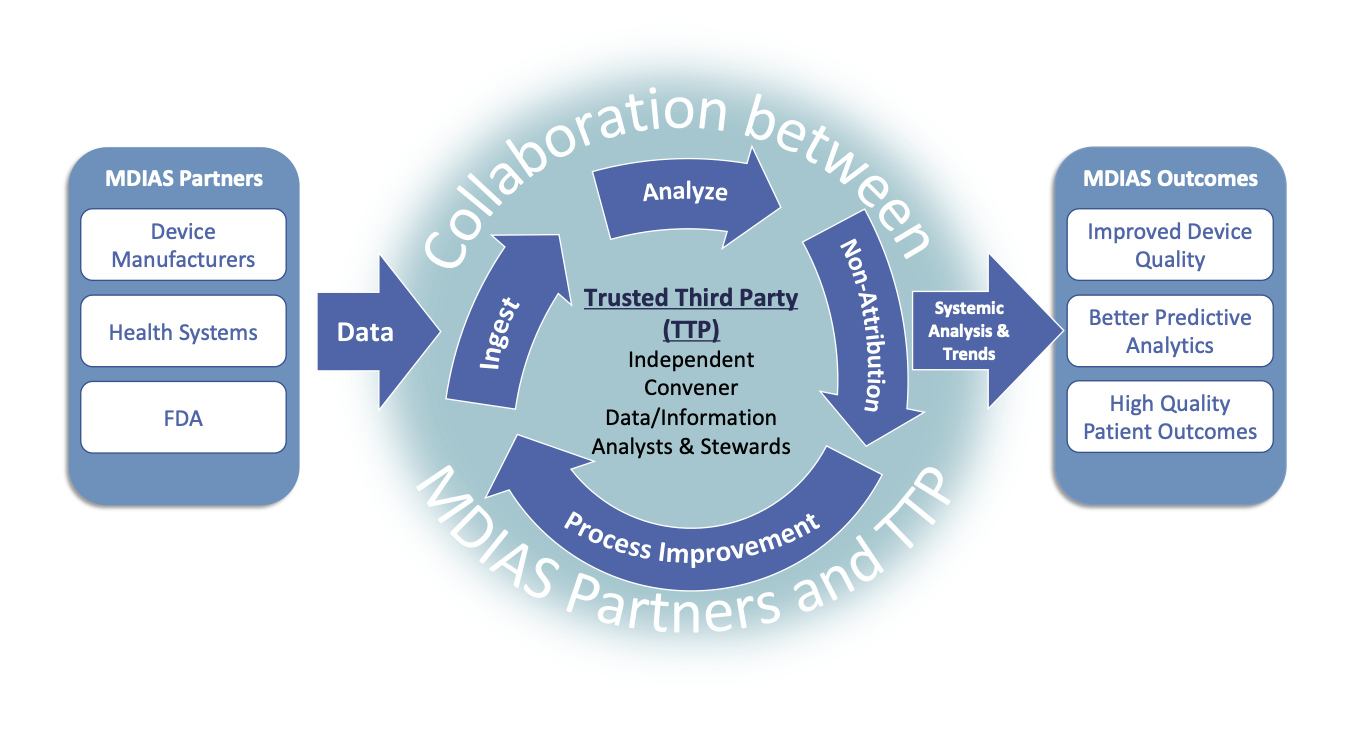
MDIAS enables us to acquire, integrate, and analyze multiple data sources in a way that provides new insights into systemic quality issues that either could not otherwise be identified or could not be identified faster than traditional methods would have allowed.
Initially, MDIAS Early Adopters include a subset of the medical device ecosystem that contribute time, energy, expertise, and data to the effort.
The intention is for MDIAS Early Adopters to become Partners and Data Providers. They will share data with the independent, Trusted Third Party, or TTP who is responsible for 1) facilitating the development of MDIAS in collaboration with the MDIAS community, as well as 2) receiving, storing, processing, and safeguarding data. The MITRE Corporation is serving as the TTP for MDIAS and has experience standing up data-sharing partnerships focused on quality and safety.
In this partnership model, the TTP works collaboratively with MDIAS members to conduct systemic analysis on the data and generate results. As more industry organizations participate, MDIAS will fuse various healthcare data sources and proactively identify quality and safety insights. These insights can be used to assess the impact of changes in the medical device ecosystem. Over time, this will benefit the entire industry and ultimately, improve people’s lives.
