MDIAS Governance
Governing Committee
The MDIAS partnership is led by a Governing Committee that consists of strategic advisors from Case for Quality and Trade Associations.

Al Crouse, MBA (Co-Chair)
Vice President of Quality & Regulatory Affairs
CVRx

Max Koenen (Co-Chair)
Associate Director – Quality
McKesson

Jen Breston, MBA
Director, GPO Complaint Management & Adverse Events
Becton Dickinson

Vishal Bhalla, MBA, MS
SVP, Enterprise Chief Experience Officer
Advocate Health

Chris Dible
Senior Director, Quality Systems
Stryker

Keisha Thomas, MS, MHS
Associate Director, Office of Product Evaluation and Quality
FDA CDRH

Sara Sulfridge
Senior Director of Strategy
Baxter Healthcare

Stephanie Fossat-Dominguez, MS
Vice President, Operations
ALTA Biomaterials [formerly known as CeloNOVA]

Rob Krhoun
(Past GC Co-Chair and Member)
Global Regulatory Compliance Manager and Internal Audit Process Steward
Boston Scientific

Michael Ruhlen, MD, MHCM, FACHE
(Past GC Co-Chair and Member)
Chief Clinical and Science Officer
LifeSpan Digital Health
[retired Atrium Health, VP, Medical Education]
MDIAS Partnership
There are currently three groups under the MDIAS Governing Committee. These include a Strategic Communications Working Group, Study 1 Working Group and Study 2 Working Group (Figure 1).
The MDIAS partnership model also includes an independent Trusted Third Party (TTP) that manages the data shared by participating partners. MITRE, a not for profit 501 c that operates Federally Funded Research and Development Centers (FFRDCs) serves as the Trusted Third Party (TTP). MITRE is responsible for:
- Facilitating the development of MDIAS in collaboration with the medical device community
- Receiving, storing, processing, and safeguarding data provided by industry partners
The TTP works collaboratively with MDIAS Partners to conduct systemic analysis on the data and generate results for MDIAS Partners to use for organizational and system-wide performance improvement.
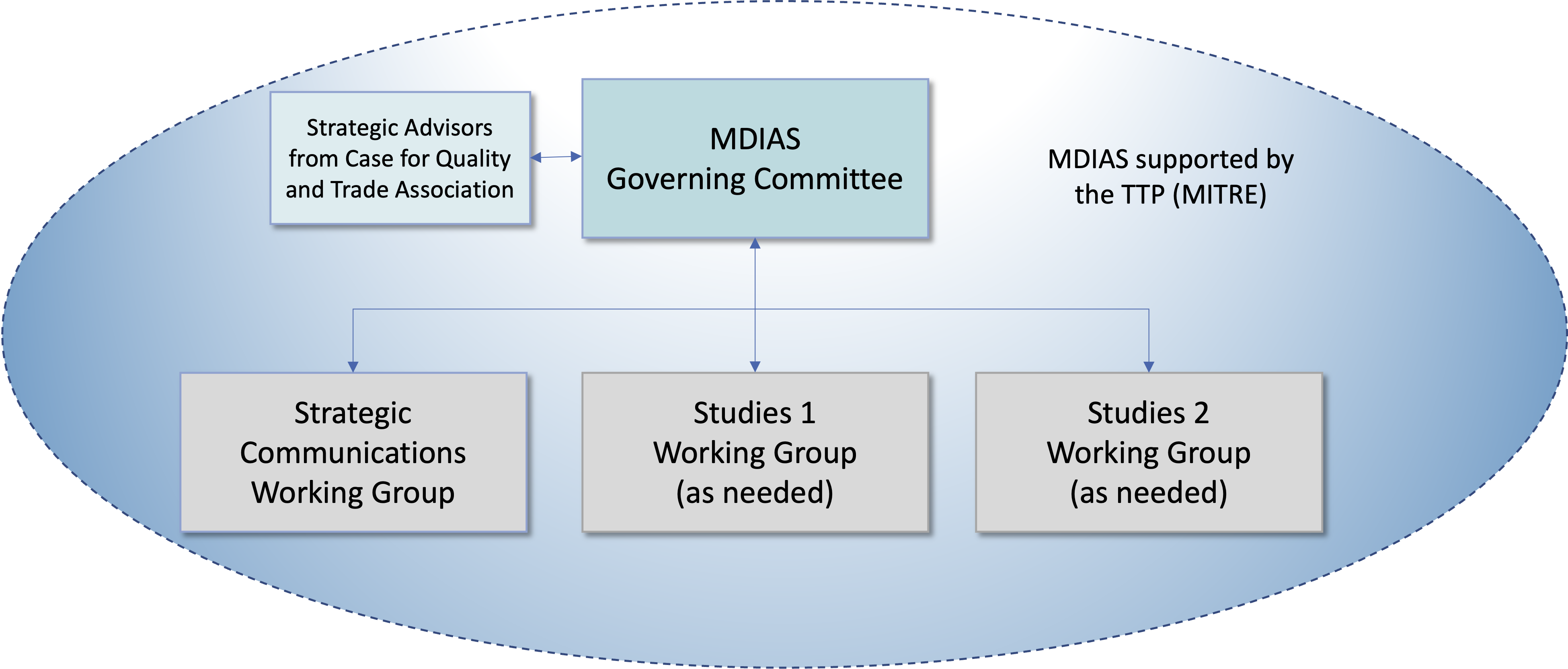
Figure 1. MDIAS Partnership Model
MDIAS Governing Committee

Figure 2. MDIAS Governing Committee
The MDIAS Governing Committee (or GC) operates as the decision-making body for MDIAS activities. GC Members set the MDIAS partnership strategy and future strategic direction, including overseeing the sustainment and growth of MDIAS and overseeing the MDIAS research studies. GC Members also act as points of contact with parties external to MDIAS and play a critical role in communicating MDIAS activities with stakeholders in the medical device safety community.
MDIAS draws GC Members from stakeholder organizations in the medical device safety community as well as the broader healthcare ecosystem. GC Members serve 2-year terms and can be re-elected once for a second 2-year term. The GC offers one seat to the U.S. Food and Drug Administration that ensures the public is represented in MDIAS.
MITRE facilitates the MDIAS Governing Committee activities by keeping members knowledgeable about MDIAS activities when members are not in committee, supporting decision-making for strategic management of MDIAS, hosting and facilitating the committee meetings themselves, and managing the administrative activities of the GC – such as scheduling, documentation, and archiving.
If you are interested in participating in the MDIAS Governing Committee, reach out to mdias_gc@mitre.org today!
