Partnership Model
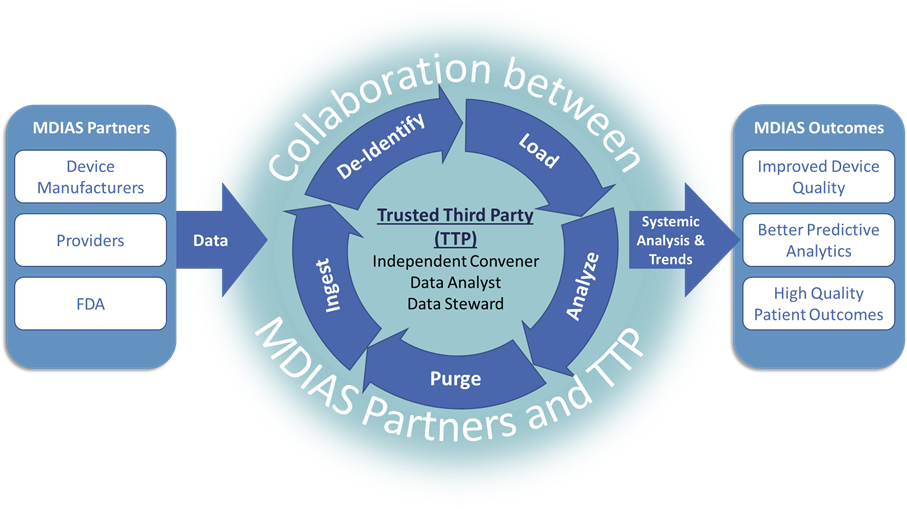
The MDIAS community currently includes a subset of the medical device ecosystem – device manufacturers, healthcare providers, and the Food and Drug Administration – that contribute time, expertise, and data to the effort. In this partnership model, these MDIAS Early Adopters share data with an independent Trusted Third Party (TTP) who is responsible for:
(1) facilitating the development of MDIAS in collaboration with the medical device community
(2) receiving, storing, processing, and safeguarding data provided by industry partners
The TTP works collaboratively with MDIAS Early Adopters to conduct systemic analysis on the data and generate results for the Early Adopters to use for organizational and system-wide performance improvement. MITRE serves as the independent TTP for MDIAS.
MDIAS Process